
Retatrutide is an investigational medication that has gained attention in ongoing research related to metabolic health, weight management, and cardiometabolic conditions. While early data has generated interest among clinicians and patients alike, it is important to understand that retatrutide is not currently approved for general medical use and remains under clinical investigation.
Retatrutide is a multi-receptor peptide therapy currently being studied for its potential effects on metabolic regulation. It belongs to a class of medications that interact with hormone pathways involved in appetite, glucose metabolism, and energy balance.
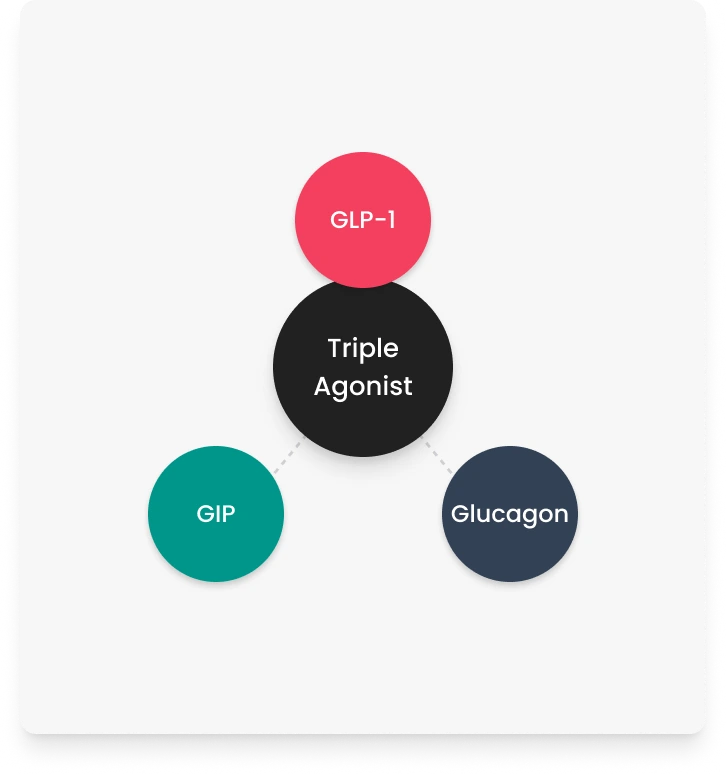
GLP-1 (glucagon-like peptide-1) receptor
Associated with appetite regulation and insulin secretion
GIP (glucose-dependent insulinotropic polypeptide) receptor
May influence insulin response and fat metabolism
Glucagon receptor
Involved in energy expenditure and glucose balance
May influence satiety signals and eating behavior through GLP-1 pathway activation.
Being studied for effects on insulin secretion and blood sugar regulation.
May influence satiety signals and eating behavior through GLP-1 pathway activation.
Retatrutide is still considered an investigational medication. It has progressed through early and mid-stage clinical trials, with larger, longer-term studies ongoing.
When multiple pathways are activated together, their effects may reinforce each other:
Early Trials
Phase I and Phase II studies completed
Mid-Stage Trials
Phase II results demonstrated potential efficacy
Ongoing Large Studies
Phase III trials currently in progress
Regulatory Review
Not yet initiated (timeline uncertain)
Current clinical trials are evaluating retatrutide for several potential therapeutic applications.
Being studied for potential effects on blood sugar regulation and glucose metabolism.
Early-stage trials have explored effects on body weight through mechanisms involving appetite, satiety, and energy expenditure. However, outcomes vary across studies, and more data is needed to understand durability and real-world applicability.
Some trials examine broader cardiovascular and metabolic outcomes.
Research includes lipid levels, blood pressure, and markers of cardiovascular risk. These endpoints are complex and require long-term data. At this stage, any conclusions remain preliminary.
We closely follow peer-reviewed research, clinical trial updates, and regulatory developments. We do not recommend or provide access to investigational medications outside of appropriate clinical and regulatory frameworks.
Safety remains the primary concern with any new therapy. Even when early data appears promising, long-term effects, rare side effects, and interactions must be thoroughly studied.
We support patient interest by providing clear, factual information, helping patients understand timelines and limitations, and offering guidance on currently available, evidence-based treatments.
Medical research evolves quickly. Following credible sources and reviewing updated clinical findings can help you stay current.
GLP-1 Medications Overview
Peptide Therapy Guide
Metabolic Health Treatments
While retatrutide is still being studied, there are existing, approved treatments that may be appropriate depending on your health goals. A licensed healthcare provider can help determine what treatments are currently available and whether they align with your needs.
If you are specifically interested in investigational therapies, clinical trials may be an option. Participation involves meeting specific eligibility criteria, undergoing structured monitoring, and understanding potential risks.

Most available data comes from relatively short- to medium-term studies. Questions remain about:
Retatrutide has not received FDA approval for general use. Availability is limited to research settings, and timelines for potential approval remain uncertain.
Like other medications in its class, retatrutide may be associated with side effects. These are still being evaluated and may include gastrointestinal symptoms or other systemic effects. The full safety profile is not yet established.
Common questions about retatrutide, answered objectively
No. Retatrutide is an investigational medication and is not approved for general use. It is currently being studied in clinical trials.
Retatrutide is designed to target three receptors (GLP-1, GIP, and glucagon), whereas many other compounds target one or two. This multi-receptor approach is a key focus of current research.
Some clinical studies have explored changes in body weight as an outcome. While results have generated interest, more research is needed to confirm findings and understand long-term effects.
Safety is still being evaluated. Current research suggests certain side effects may occur, but long-term safety data is not yet fully available.
This is still being studied. Clinical trials typically focus on specific populations, and broader applications—if any—would depend on future research and regulatory review.
There is no confirmed timeline. Approval, if pursued, would depend on the outcomes of ongoing and future clinical trials.